 CASE
REPORT
Congenital leukemia after heavy abuse of permethrin
spray during pregnancy
A Borkhardt, M
Wilda, U Fuchs, L
Gortner and I Reiss
Children’s University Hospital Giessen, Feulgenstr 12, 35392
Giessen, Germany
Correspondence to:
Dr Borkhardt, Paediatric
Haematology and Oncology, Feulgenstr 12, 35392 Giessen, Germany;
arndt.borkhardt{at}paediat.med.uni-giessen.de
Accepted 6 October 2002
ABSTRACT
A single case is described of
congenital leukemia with 11q23/MLL rearrangement in a preterm female
newborn. Because of arachnophobia, the mother had heavily abused
aerosolized permethrin, a widely used household insecticide.
Permethrin is considered comparatively safe, but, in view of the
mother’s history, its potential to induce cleavage of the MLL gene in
cell culture was tested. Incubation of the BV173 cell line with 50 µM
permethrin readily induced MLL cleavage.
Keywords: congenital leukemia; leukemia; permethrin; insecticide;
11q23/MLL
Congenital leukemia is a rare disease which affects about one
child in 200 000–250 000 live births a year. In spite of its
rarity, congenital leukemia has stimulated much interest because of
the opportunity to learn more about the underlying causes of in utero
leukaemogenesis. There are many risk factors that may increase the
chance of early induction of a leukaemic phenotype—for example,
parental exposure to occupational and environmental toxins, use of
tobacco, marijuana, alcohol, and other toxins.1
Congenital and infant leukemia are often associated with
rearrangements of the MLL gene at chromosome 11q23.2
Several substances have been examined for their potential to cross
the placental barrier and to induce such a chromosomal break at
11q23/MLL.3,4
Here we extend the list of drugs capable of cleaving the MLL gene,
at least when cells are exposed in culture. Together with a rather
unusual case history, our experimental data shed some light on the
generation of congenital leukemia and may help to prevent some of
the fatal cases of this disease.
CASE
REPORT
A 27 year old healthy woman was admitted to the
gynecology department because of premature labor. She was in the
35th week of her first pregnancy. Prenatal check ups had been
normal. As the cardiotocogram showed no fetal heart rhythm, an
emergency caesarean section was planned. However, six minutes later,
the woman spontaneously gave birth to a premature infant
weighing 2300 g and showing no vital signs but a striking livid
color of the skin. Primary cardiopulmonary resuscitation was
started immediately with endotracheal intubation, chest
compression, and repetitive administration of adrenaline. However,
the Apgar score remained 0 after five and 10 minutes. The pediatric
emergency team arrived 10 minutes later and continued to perform
cardiopulmonary resuscitation. A central line was achieved by
catheterization of the umbilical vein. At 25 minutes post partum, a
stable circulation was established, with a heart rate of 140
beats/min. A physical examination showed hepatomegaly of 5 cm below
costal margin and nodular livid infiltrations of the skin (fig 1A
below).
|
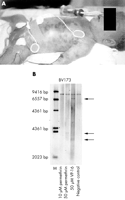 |
Figure 1 (A) The patient at 2 hours
of age. Note the leukaemic skin infiltration ("blueberry spots").
(B) Southern blot showing an MLL rearrangement in the BV173 cell
line incubated with 50 µM permethrin for six hours. VP16 induced MLL
cleavage was used as a control. |
Apart
from normal anatomy of the heart, the ultrasound showed substantial
pulmonary hypertension combined with second to third degree
insufficiency of the tricuspid valve. Besides hepatomegaly, a
bilateral intracranial hemorrhage was found. For sufficient
oxygenation and carbon dioxide removal, high frequency ventilation
was needed with 10 Hz, FIO2 = 1, and a
mean airway pressure of 13 cm H2O. The initial white blood
cell count was 400 000/µl with an excess of myeloid blast cells.
Examination of the bone marrow confirmed the diagnosis of a
congenital acute myeloid leukemia, with 95% blast cells of
myelomonocytic morphology (FAB-M5). A complete blood exchange was
performed, which reduced the white blood cell count to 56 000/µl and
resulted in an improvement in oxygenation and ventilation. Twelve
hours later the white blood cell count had increased again, to
125 000/µl. Extracorporeal membrane oxygenation was considered
but eventually rejected because of the progressive intracranial
hemorrhage involving a large area of the parenchyma. The infant
died 48 hours after birth from multiorgan failure. Apart from
the severe brain damage already diagnosed by ultrasound, the
autopsy showed diffuse infiltration of lungs, liver, spleen,
and cutis.
CELL CULTURE
EXPERIMENTS: INDUCTION OF MLL REARRANGEMENTS BY
PERMETHRIN
The experimental assay was performed exactly as
described by Strick et al,3
who analyzed a wide range of natural substances in food and dietary
supplements for their ability to induce site specific cleavage within
the MLL gene.
RESULTS AND
DISCUSSION
Cytogenetic analysis of the bone marrow aspirate
showed the presence of a translocation t(11;19)(q23;p13) in all
metaphases analyzed. Furthermore, we showed a rearrangement of the
MLL oncogene at chromosome 11q23 by Southern blotting. Routine
immunophenotyping showed expression of CD15, CD33, CD 65s, and MPO
(data not shown). In previous studies, the monoclonal antibody 7.1
emerged as a valuable immunophenotypic tool for the detection of
leukaemic cells, with NG2 antigen expression which correlates
strongly with MLL rearrangements in infants.5,6
In our case, however, staining with this monoclonal antibody remained
negative, which prevented us from carrying out further cell sorting
experiments.
The mother had apparently suffered from arachnophobia since early
childhood. Two years before her pregnancy she began to overuse
aerosolized permethrin. Since then, she had lived alone. People
avoided visiting her because of the penetrating odor in her house.
Permethrin is a widely used household insecticide providing
protection from the malaria vector Anopheles.7,8
It is also known to be an efficacious drug against scabies and
the head louse Pediculosis capitis.9,10
Compared with other drugs, it is considered safe and its topical use
is recommended even for neonates with scabies.11
However, it has been linked to the generation of both chromosomal
aberrations in bone marrow cells of mice and DNA lesions in human
lymphocytes.12–14 We therefore hypothesized that permethrin crossed the placenta
of the pregnant woman and affected the haematopoietic precursor
cells in the developing fetus, resulting in leukaemogenesis. We
therefore tried to generate the rearrangements within the MLL
oncogene in vitro by exposing BV173 cells to 50 µM permethrin. After
24 hours of exposure, the permethrin treated BV173 cells showed a
clear MLL rearrangement, whereas the untreated cells showed the wild
type MLL gene only (fig 1B above). Therefore we strongly recommend great caution
in the use of permethrin during pregnancy. Even if definitive
conclusions cannot be drawn from a single isolated case, our data
strongly suggest that permethrin may have severe side effects when
fetal haematopoietic precursor cells are exposed in utero. In the
same vein, a recent report from a multinational collaboration
indicated that use of mosquitocidal drugs during pregnancy is
significantly associated with infant leukaemia.4
It is especially noteworthy that the authors found this association
only for the MLL rearranged cases and not for MLL germline cases.
This strongly argues against selection bias in their case-control
study and supports the hypothesis that in utero exposure causes the
MLL rearrangements.
Finally, the mother in our case was successfully treated for
arachnophobia by psychotherapy. Two years later, she gave birth
to a healthy term boy who had no clinical sign of leukemia and
who is still healthy at the age of 13 months. ACKNOWLEDGEMENTS
Expert technical assistance
from Claudia Keller and Stefanie Garkisch is gratefully acknowledged.
We thank Jochen Harbott, Giessen, Germany for providing us with the
cytogenetic data. Immunophenotypic data were kindly provided by W-D
Ludwig, Robert Rössle Cancer Centre, Humboldt University, Berlin,
Germany. The molecular studies were supported by the German Cancer
Foundation (grant 10-1658-Bo2).
REFERENCES
- Sande JE, Arceci RJ, Lampkin BC. Congenital and neonatal
leukemia. Semin Perinatol 1999; 23:274–85.[Medline]
- Ford AM, Ridge SA, Cabrera ME, et al. In utero
rearrangements in the trithorax-related oncogene in infant leukaemias.
Nature 1993;363:358–60.[CrossRef][Medline]
- Strick R, Strissel PL, Borgers S, et al. Dietary
bioflavonoids induce cleavage in the MLL gene and may contribute to infant
leukemia. Proc Natl Acad Sci USA 2000;97:4790–5.[Abstract/Free Full Text]
- Alexander FE, Patheal SL, Biondi A, et al.
Transplacental chemical exposure and risk of infant leukemia with MLL gene
fusion. Cancer Res 2001;61:2542–6.[Abstract/Free Full Text]
- Smith FO, Rauch C, Williams DE, et al. The human
homologue of rat NG2, a chondroitin sulfate proteoglycan, is not expressed on
the cell surface of normal hematopoietic cells but is expressed by acute
myeloid leukemia blasts from poor-prognosis patients with abnormalities of
chromosome band 11q23. Blood 1996;87:1123–33.[Abstract/Free Full Text]
- Behm FG, Smith FO, Raimondi SC, et al. Human
homologue of the rat chondroitin sulfate proteoglycan, NG2, detected by
monoclonal antibody 7.1, identifies childhood acute lymphoblastic leukemias
with t(4;11)(q21;q23) or t(11;19)(q23;p13) and MLL gene rearrangements.
Blood 1996;87:1134–9.[Abstract/Free Full Text]
- Rowland M, Durrani N, Hewitt S, et al.
Permethrin-treated chaddars and top-sheets: appropriate technology for
protection against malaria in Afghanistan and other complex emergencies.
Trans R Soc Trop Med Hyg 1999;93:465–72.[Medline]
- Binka FN, Mensah OA, Mills A. The cost-effectiveness of
permethrin impregnated bednets in preventing child mortality in
Kassena-Nankana district of Northern Ghana. Health Policy
1997;41:229–39.[CrossRef][Medline]
- Blanshard ME, Schofield J. Scabies: a practical approach.
Prof Care Mother Child 1999;9:15–16.[Medline]
- Dodd CS. Interventions for treating headlice. Cochrane
Database Syst Rev 2000;CD001165.
- Quarterman MJ, Lesher JL, Jr. Neonatal scabies treated
with permethrin 5% cream. Pediatr Dermatol 1994;11:264–6.[Medline]
- Cantalamessa F. Acute toxicity of two pyrethroids,
permethrin, and cypermethrin in neonatal and adult rats. Arch Toxicol
1993;67:510–13.[Medline]
- Barrueco C, Herrera A, Caballo C, et al. Induction
of structural chromosome aberrations in human lymphocyte cultures and CHO
cells by permethrin. Teratog Carcinog Mutagen 1994;14:31–8.[Medline]
- Santoni G, Cantalamessa F, Spreghini E, et al.
Alterations of T cell distribution and functions in prenatally
cypermethrin-exposed rats: possible involvement of catecholamines.
Toxicology 1999;138:175–87.[CrossRef][Medline]
|